Synthetic polymers in most cases do not have one bespoke molecular weight. A sample typically consists of a large number of individual polymer chains, each having a different molecular weight. The average molecular weights and the shape of the molecular weight distribution are a kinetic fingerprint of how to polymer material was made. The resulting molecular weight distribution dictates physical and mechanical properties.
In free radical polymerizations, four key mechanistic events need to be considered. These are initiation, propagation, termination, and chain transfer. The latter often gets brushed under the carpet in introductory textbooks, but is pivotal.
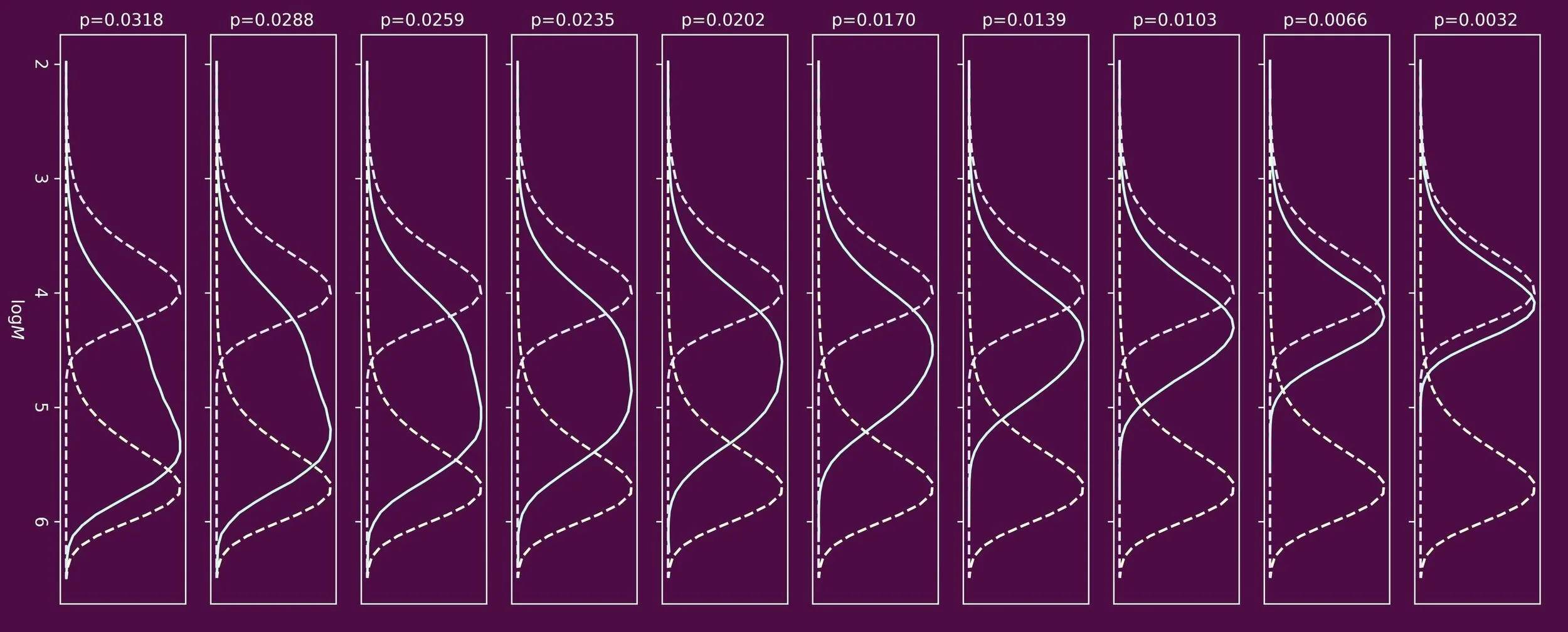
When one targets polymers of low molecular weight, chain transfer agents are often used. One prominent class of chain transfer agents are thiol compounds, for example n-dodecanethiol. To understand how the molecular weight distribution develops throughout the polymerization process, the ability to determine the reactivity of the chain transfer agent is crucial. This reactivity is often expressed in the form of a chain transfer constant, Ctr, which is the ratio of the rate coefficients of chain transfer and propagation.